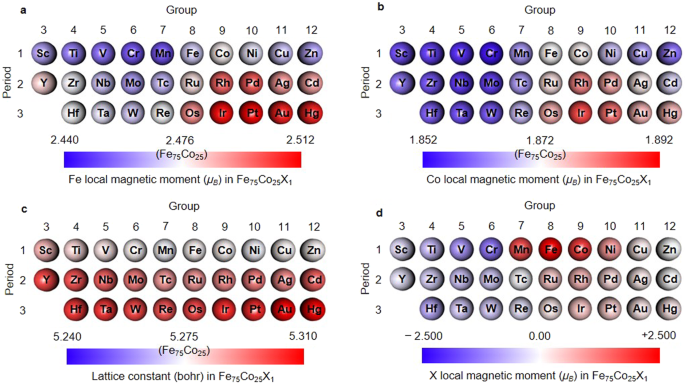
Machine learning autonomous identification of magnetic alloys beyond the Slater-Pauling limit | Communications Materials

The movement of momentum accompanying magnetic moment in iron and nickel: Stewart, John Quincy: Amazon.com: Books

SOLVED:(II) An iron atom has a magnetic dipole moment of about 1.8 ×10^-23 A ·m^2 (a) Determine the dipole moment of an iron bar 9.0 cm long, 1.2 cm wide, and 1.0
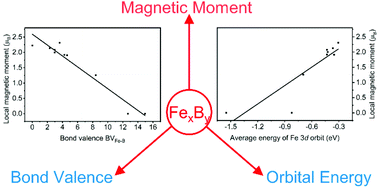
Insight into the magnetic moment of iron borides: theoretical consideration from the local coordinative and electronic environment - Dalton Transactions (RSC Publishing)

Density functional theory study of the magnetic moment of solute Mn in bcc Fe | Mark Wenman Research Group
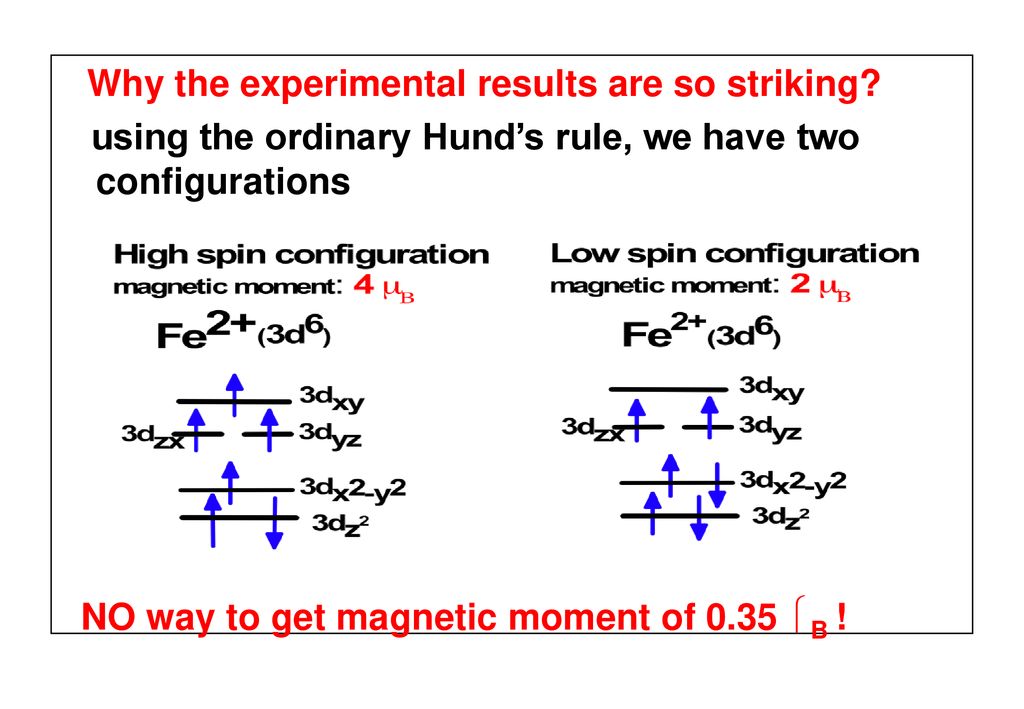
Theory of Magnetic Moment in Iron Pnictides (LaOFeAs) Jiansheng Wu (UIUC), Philip Phillips (UIUC) and A.H.Castro Neto (BU) arxiv: ppt download

A review of high magnetic moment thin films for microscale and nanotechnology applications: Applied Physics Reviews: Vol 3, No 1
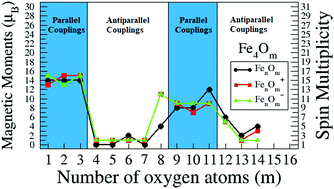
Relation between structural patterns and magnetism in small iron oxide clusters: reentrance of the magnetic moment at high oxidation ratios - Physical Chemistry Chemical Physics (RSC Publishing)

SOLVED:The magnetic dipole moment of the iron atom is about 2.1 ×10^-23 A ·m^2 . (a) Calculate the maximum magnetic dipole moment of a domain consisting of 10^19 iron atoms. (b) What
![A complex ion [Fe(H2O)6]^2 + is found to be paramagnetic. Its magnetic moment is 4.89 BM. How many unpaired electrons are there? A complex ion [Fe(H2O)6]^2 + is found to be paramagnetic. Its magnetic moment is 4.89 BM. How many unpaired electrons are there?](https://d1hj4to4g9ba46.cloudfront.net/questions/1560204_1727112_ans_6b23900fef074fa28cabacf524568d21.jpg)


![The spin magnetic moment of iron in K(3)[Fe(CN)(6)] The spin magnetic moment of iron in K(3)[Fe(CN)(6)]](https://doubtnut-static.s.llnwi.net/static/web-thumb/644377997_web.png)
![The spin magnetic moment of iron in K3 [ Fe (CN)6 ] is : The spin magnetic moment of iron in K3 [ Fe (CN)6 ] is :](https://dwes9vv9u0550.cloudfront.net/images/4561692/4493ffc6-001d-4396-8ed9-3c26b6a9e01b.jpg)


![The spin magnetic moment of iron in K(3)[Fe(CN)(6)] The spin magnetic moment of iron in K(3)[Fe(CN)(6)]](https://d10lpgp6xz60nq.cloudfront.net/ss/web/1269215.jpg)

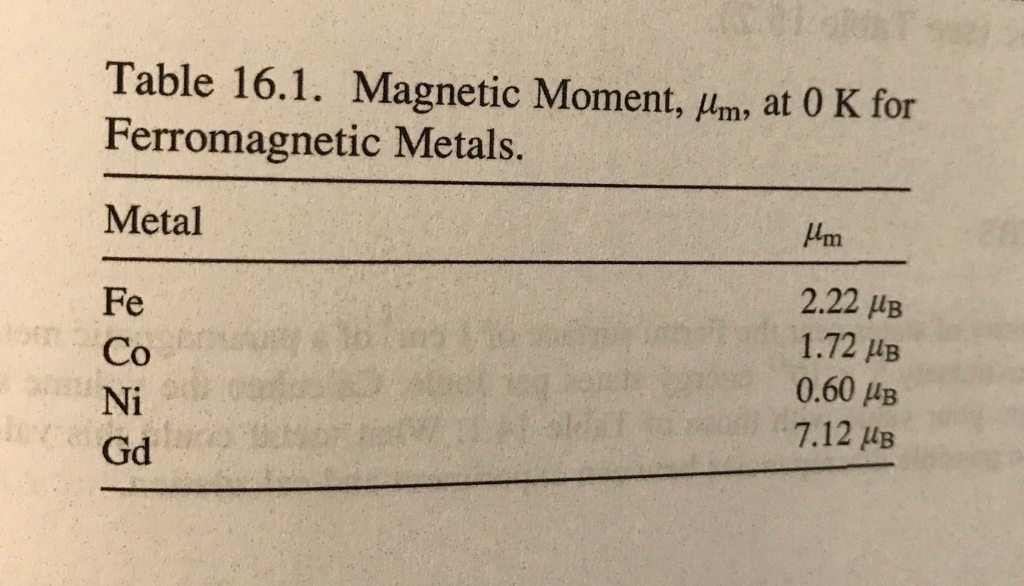


![The spin magnetic moment of iron in K3 [ Fe (CN)6 ] is : The spin magnetic moment of iron in K3 [ Fe (CN)6 ] is :](https://haygot.s3.amazonaws.com/questions/1952807_1117559_ans_880cbb7d24bb4d01ab49a890bc90ed14.jpg)

![The spin magnetic moment of iron in `K_(3)[Fe(CN)_(6)]` - YouTube The spin magnetic moment of iron in `K_(3)[Fe(CN)_(6)]` - YouTube](https://i.ytimg.com/vi/m2ePFFONgdA/maxresdefault.jpg)